Ethical Dimensions of STEM Research
Introduction to the Module
In this module, you will hear from Dr. Nancy Tuana, Director of the Rock Ethics Institute and Professor of Philosophy at the Pennsylvania State University and Dr. Thomas Richard, Director of the Penn State Institutes for Energy and the Environment and Professor of Agricultural and Biological Engineering at the Pennsylvania State University. Drs. Tuana and Richard will present an introduction to the ethical dimensions of STEM research, including research integrity, embedded ethics, and broader impacts. You will have opportunities to test your knowledge as we go.
By the end of this module, you will be able to:
- List the ethical dimensions of STEM research
- List examples and questions one should ask one’s self in the course of conducting research for each dimension
- Identify ethical dimensions present in STEM research scenarios
Introduction to the Ethical Dimensions of STEM Research
Working with scientists and engineers has helped us appreciate the relevance of ethics to many aspects of the work done by STEM researchers. This video provides an introduction to a broader model of the ethical dimensions of research in STEM fields, including research ethics, broader impacts, and embedded ethics.
Research Integrity
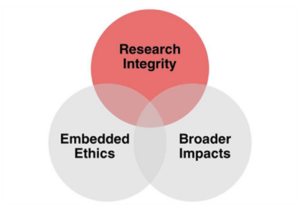
What do we mean by “Research Integrity”?
There are the ethical considerations of how to proceed in the course of conducting research in science and engineering. These concepts are termed “research integrity.” The National Institutes for Health, for example, refers to research integrity as including the following:
- the use of honest and verifiable methods in proposing, performing, and evaluating research
- reporting research results with particular attention to adherence to rules, regulations, guidelines, and
- following commonly accepted professional codes or norms.
Our conception of Research Integrity is consistent with the conception of Responsible Conduct of Research (RCR) set forth by the National Office of Research Integrity (ORI). In The ORI Introduction to the Responsible Conduct of Research, Nicholas Steneck explains that:
“…expectations for the responsible conduct of research are complex and not always well defined… Some responsible practices are defined through law and institutional policies that must be followed. Others are set out in non-binding codes and guidelines that should be followed. Still other responsible practices are commonly accepted by most researchers but not written down. Instead, they are transmitted informally through mentoring, based on the understandings and values of each mentor.”
For this reason, understanding what is involved in conducting research responsibly is an important component of research integrity.
Many universities and research organizations now require RCR training. Research misconduct is one important domain of RCR training.
Of all of the areas in research integrity, research misconduct may be the most familiar. Research misconduct, namely, fabrication, falsification, and plagiarism, involve research choices made by a scientist or engineer where the act of wrongdoing is often the most clear and in which there is little ethical ambiguity. One can certainly fabricate or falsify more or less data, or plagiarize a little or a lot, but just the mere fact of doing so makes the action a violation of the basic ethical expectation of responsible research.
The ORI defines Research Misconduct as follows:
Research misconduct means fabrication, falsification, or plagiarism in proposing, performing, or reviewing research, or in reporting research results.
- Fabrication is making up data or results and recording or reporting them.
- Falsification is manipulating research materials, equipment, or processes, or changing or omitting data or results such that the research is not accurately represented in the research record.
- Plagiarism is the appropriation of another person’s ideas, processes, results, or words without giving appropriate credit.
- Research misconduct does not include honest error or differences of opinion.
Plenty of examples of research misconduct exist. The following pages discuss two from different disciplines that may be familiar to you.
Link between autism and vaccines
The work of Andrew Wakefield, which initially posited a link between autism and vaccines, was found to contain falsified research data. This work has had drastic implications, including the promotion of anti-vaccine communities, which may have in turn caused the resurgence of previously eradicated diseases. Here is a timeline of the case. This is the original paper.
Organic Semiconductors
While at Bell Labs, Jan Hendrik Schön produced cutting-edge research on organic semiconductors at a rapid rate. When several scientists in the community raised questions about the validity of his data, a committee was convened, and according to an article in the New York Times, they found that “that data in the disputed research, published between 1998 and 2001, had been improperly manipulated, even fabricated, confirming suspicions raised by outside scientists in May. The committee placed the blame for the deceit on one Bell Labs scientist, Dr. J. Hendrik Schön.” Furthermore, according to the same article, “Dr. Schön told the committee he had deleted almost all of the original data files because his computer lacked hard disk space to store the files. He said he had no laboratory notebooks. Dr. Schön also could not reproduce any of the findings for the committee.” If you are interested in further details of the Schön case, we recommend Plastic Fantastic: How the Biggest Fraud in Physics Shook the Scientific World by Eugenie Samuel Reich.
Grey Areas of Research Misconduct
Even in the domain of fabrication, falsification, and plagiarism, there are some grey areas in which the actions are questionable, yet the wrongness of the activity is not clear-cut. Consider the case where a faculty member uses text from a paper he or she has co-authored with a graduate student in a publication or a grant application, but does not ask the student’s permission to do so. It might even be the case that the faculty member gave the student appropriate authorship credit. But if we use another person’s work in another venue, are we obligated to first get that person’s permission to do so? What if the text cited could lead to others “scooping” the graduate student’s insights before she or he is able to publish the results of the research?
These grey areas have been identified by some as “Questionable Research Practices.” The Vlaams Instituut voor Biotechnologie (VIB), a life sciences research institute located in Flanders, Belgium lists the following examples of Questionable Research Practices:
- Neglecting negative outcomes
- Using inappropriate statistics to support one’s hypothesis
- Inappropriate research design
- Leaving out relevant controls
- Inappropriate re-use of controls
- Removal of ‘outliers’
- Conscious bias
- Unethical experimentation
- Peer review abuse
We know it is wrong to use fake data or steal another’s ideas and claim them to be their own. But what about more complex issues that are beyond blatant research misconduct?
Responsible Conduct of Research: Beyond Research Misconduct
RCR training involves recognition not only of violations of research integrity, such as research misconduct due to falsification or fabrication of data and plagiarism, but also the knowledge and values needed to respond to the grey areas of research integrity. You can read more about each of these areas of research integrity in The ORI Introduction to the Responsible Conduct of Research.
Human Subjects
Transcript:
“So another set of-another domain in responsible conduct of research has to do with the treatment of research subjects, and that falls into two domains. One is research on human subjects, and the other is research on animal subjects. They’re somewhat different, but in both cases a general principal underlying both of them is the principal of doing no harm. Now that’s a basic principal in ethics-in particular, in bioethics-that deals with either research on human subjects or health practices on humans, and means that we shouldn’t be causing people harm that isn’t in someway counteracted by a benefit that they will receive. With humans that principal gets applied somewhat differently than it does with animal subjects, and the reason is the principal of moral standing with humans. We actually think as a community that humans have a different or higher moral standing than animals. And part of that entails the view that humans have a right to autonomy, and that means they have a right to informed consent. That you cannot do certain things to humans without them knowing what you’re doing and agreeing to doing it, and in particular that has to do with protection of their bodily integrity as well as their health. That means that anytime you do research and are doing it on a human subject, you have to be very careful. Not only that the work will benefit that individual, but that individual is fully informed of the nature of the research, any potential risks or harms that might come to them. As well as giving a sense of how that research might benefit either themselves or the broader society.”
Publication Practices and Responsible Authorship
Transcript:“Another domain is publication practices and responsible authorship, and there are a number of issues that fall under this domain, but I want to talk about just a few here today. One is authorship order. When you’re collaborating with other scientists or engineers, or social scientists and humanists, it’s important that the authorship credit be-reflect the role each person played. What that means is that it’s important to establish clearly and very early in the research practice who will play which role, and what those roles mean. There’s no rule of thumb for authorship order because in some of the sciences, being first author means that you put in the majority of work for the paper. But in other sciences, being the last author signifies that particular role. Most important is to understand the values and meanings of authorship order in the practice of science and engineering you’re involved in, and making sure, before a research project even starts-before you even start to write a paper or a grant proposal-that credit for each person involved is clearly established and agreed upon. “
Animal Resources
Transcript:“Research on animal subjects also resides under the value of respect, in this case, for non-human life forms. And our attitudes to research on animals has to do with our attitudes about what constitutes moral standing. Moral standing is a concept that we often we see not as either you have it or you don’t have it, but to some extent is on a continuum. We see human beings, for example, as having moral standing, and then have a continuum from humans to other life forms in terms of their moral status. Some. for example, would say that one characteristic that does give moral salience is the ability to feel pain as well as the ability to think. So higher forms of intellectual ability-so primates, for example, who are able clearly to engage in-not only do they experience pleasure and pain, they clearly are capable of relatively sophisticated, intellectual understandings-are seen as having, for example, more moral standing than a plant, like a tree or a flower. And therefore we think that the higher one’s moral standing, the more care we have to take in the animal subjects research. So protections have to do with not causing unnecessary pain and also a sustainability in a sense, a stewardship of the subjects that we’re doing research on. So that we’re not unnecessarily using animal subjects when a topic has already been explored, or using an animal subject when we could do a different kind of subject. Whether it be a modeling or a prediction instead of an animal subject.”
Mentor/Trainee Responsibilities
Transcript:
“Research on animal subjects also resides under the value of respect, in this case, for non-human life forms. And our attitudes to research on animals has to do with our attitudes about what constitutes moral standing. Moral standing is a concept that we often we see not as either you have it or you don’t have it, but to some extent is on a continuum. We see human beings, for example, as having moral standing, and then have a continuum from humans to other life forms in terms of their moral status. Some. for example, would say that one characteristic that does give moral salience is the ability to feel pain as well as the ability to think. So higher forms of intellectual ability-so primates, for example, who are able clearly to engage in-not only do they experience pleasure and pain, they clearly are capable of relatively sophisticated, intellectual understandings-are seen as having, for example, more moral standing than a plant, like a tree or a flower. And therefore we think that the higher one’s moral standing, the more care we have to take in the animal subjects research. So protections have to do with not causing unnecessary pain and also a sustainability in a sense, a stewardship of the subjects that we’re doing research on. So that we’re not unnecessarily using animal subjects when a topic has already been explored, or using an animal subject when we could do a different kind of subject. Whether it be a modeling or a prediction instead of an animal subject.”
Mentor/Trainee Responsibilities
Transcript:
“-of responsible conduct of research has to do with data management, ownership, and sharing. And this is a domain that links back in the management and recording to concerns around trustworthiness and honesty in the practice of science. One of the reasons why keeping good lab books is so important is because it’s through careful recordings of experiments and work that’s being done that scientists are, and engineers are, able to actually trace the pedigree of ideas. And, again, ensure that they’re properly attributing the sources of ideas. So part of the reason for data management and good data recording is to ensure that you don’t engage in something that verges on accidental plagiarism. In other words, attributing an idea that you got from someone else in the lab to another person. But it’s also a way to be able to document that you followed the line of research in a consistent way. To be able to go back and see where there might have been gaps or errors in the analysis and thus be able to check where you might have gone awry, if you or someone else cannot replicate the data. Data-sharing is another domain in data management recording and ownership, and sharing, which has to do with the broader impacts criteria of benefiting the wider scientific community. As scientists and engineers start to develop data-management systems or codes or such that they base their results on, at a certain point it’s important to be able to share that data with other scientists and engineers. Both so that they can replicate the work and ensure that it’s accurate, but also so they can build on the work. The tension between data-sharing is the point at which that work needs to be shared. In other words, when is a scientist or engineer sufficiently done with working in developing the data? And when is it ready to be shared with others who can then build on that work, and publish based on it? And that’s a complicated question that requires careful analysis, particularly in todays world of the Internet and the phenomena of tremendous amount of data and codes available on the Internet. “
Mentor/Trainee Responsibilities
Transcript:
“-domain of responsible conduct of research is the domain of collaboration, and in science and engineering both. Whether in the academic world or in the professional world, there’s a substantial amount of collaboration that happens in research and practice. The values that underlie responsible conduct of research with collaboration is fairness, responsibility, and accountability. When you’re the member of a research team, it’s important to know what your responsibilities are, and then to fulfill those responsibilities fully and to your best ability. Or if for some reason you’re unable to, to inform the team of that fact. The collaborate team relies on one another and relies on each partner being responsible and accountable to the others. If you’re in a collaborative team it’s very important to be clear about what each person’s responsibility is. Sometimes problems come in collaboration because people don’t all have the same understanding of their responsibilities. Whether that miscommunication is because of different expectation in the different disciplines of science or because of cultural differences due to where one was trained. Communication is key to ensuring fair, accountable, and responsible collaborative research. “
Mentor/Trainee Responsibilities
Transcript:
“–topic in responsible conduct of research, is avoiding or managing, conflicts of interest. The values underlying conflicts of interest is a concern that, they’re not be unethical bias of scientific or engineering research. In other words, if there are particular conflicts that an individual might have–for example, the work is being funded by a company who is paying that scientist large sums of money, but will pull the funding if the results are negative. That’s a potential conflict of interest that might result in the scientists leaning in the direction of developing the positive results. Maybe not even intentionally but without even being fully aware of how the questions are being structured, or the data is being analyzed. In order to avoid conflicts of interests, it’s important that they be well managed. It’s in the domain of science–once again, going back to issues of trust. Even apparent or perceived conflicts of interest can erode society’s trust in science and engineering. And thus, one of the things that’s very important to responsible conduct of research is to ensure that where possible, we avoid conflicts of interests that will bias our work. And if we have manageable conflicts of interests, that they’re well managed by an outside party. Usually they’re managed by an institutional review board, or IRB’s office, who make sure that there’s little worry about a conflict of interest.”
Mentor/Trainee Responsibilities
Transcript:
“-when relevant previous publications…peer review. Peer review? Yeah. Okay! Yikes… There are also research ethics issues surrounding the practice of peer review. Scientists and engineers-in fact, all scholars-do a substantial amount of work in peer reviewing. Both for juried publications in journals as well as for grants, as well as tenured promotion cases for academic scientists and engineers. The principals underlying peer review is objectivity and fairness. So, for example, if we think about objectivity, we have to avoid conflicts of interest. If, for example, the person who’s work you’ve been asked to review was a previous student, or is a colleague who’s work you’ve collaborated on. Or happens to be someone who for personal reasons you have animosity towards in ways you feel you cannot manage. It’s imp–“
Finally, we’d like to note that it is becoming common to discuss issues of intellectual property when discussing research integrity. Often, these issues are covered in graduate student handbooks or policies. Here at Penn State, this would mean documents like IP01-Ownership and Management of Intellectual Property and IP02-Co-Authorship of Scholarly Reports, Papers and Publications, but your university may have different policies surrounding intellectual property, so it is important to do your due diligence and inform yourself of the intellectual property rights and regulations of your institution. Additional information can be found in the “Intellectual Property Agreements” section of the RCR Administrators website and the “Data Ownership” section of the Responsible Conduct in Data Management website.